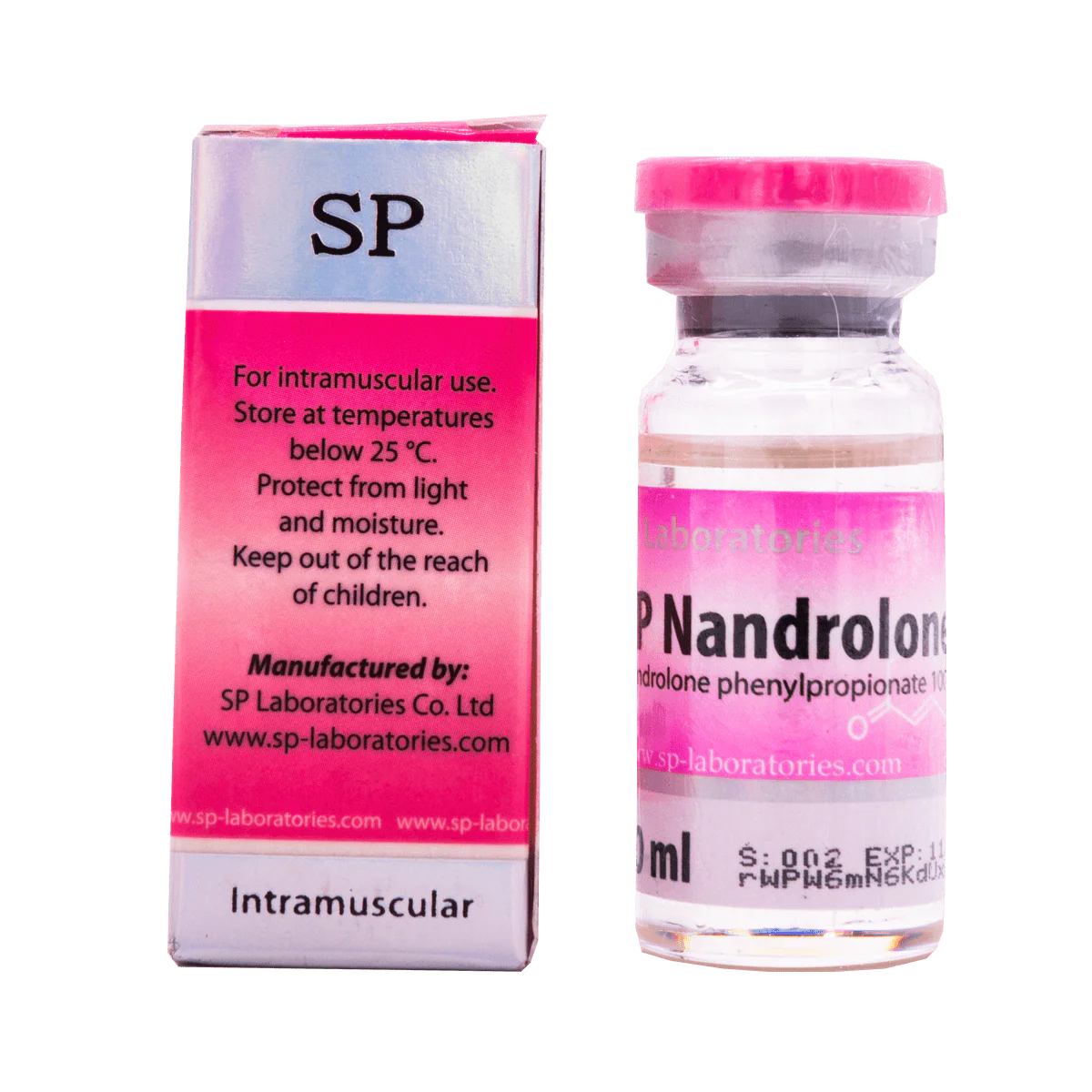
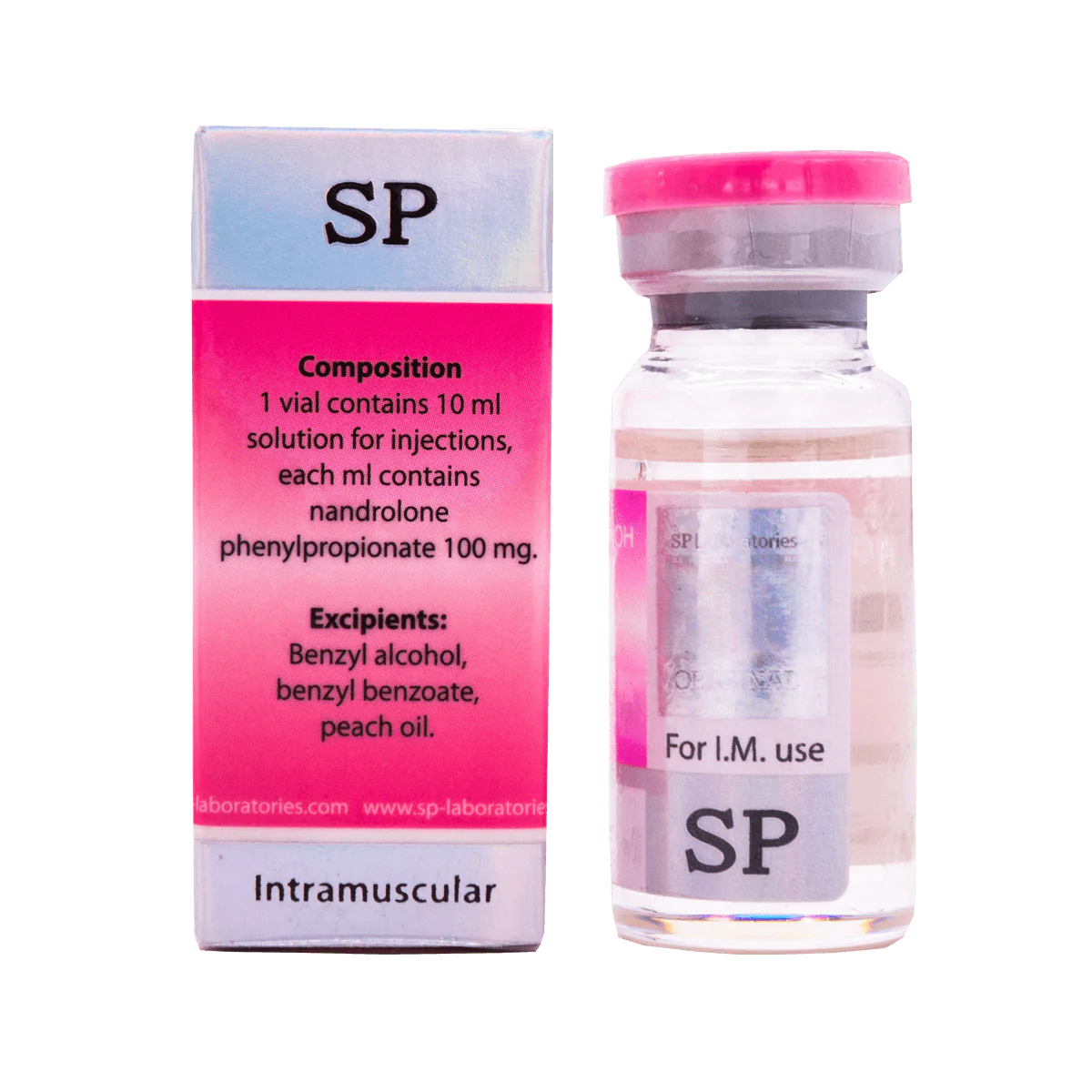
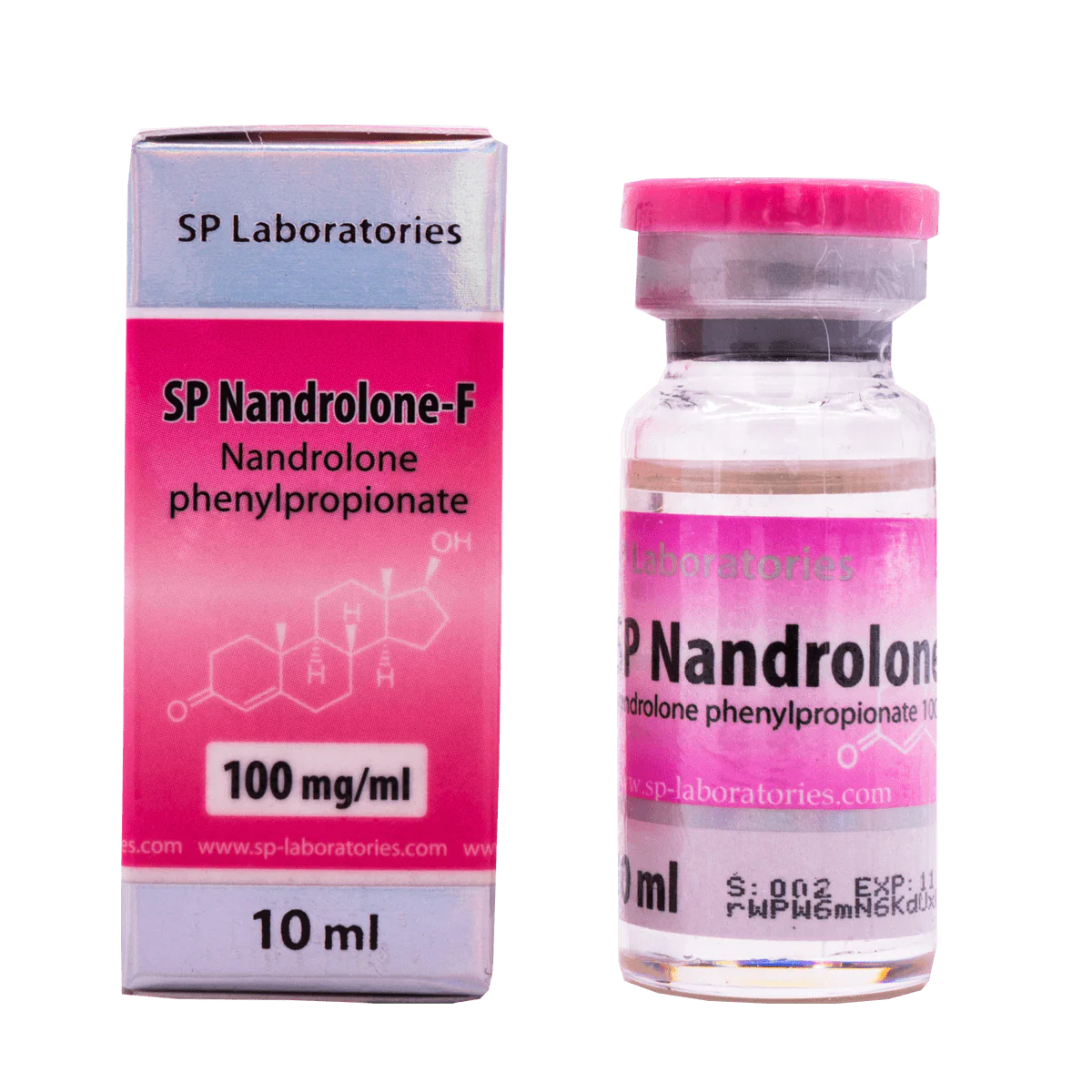
SP Labs Nandrolone F 10 ml contains nandrolone phenylpropionate as active substance in the group of anabolic steroids. This drug is very similar to nandrolone decanoate, but it does not contain durabolin, therefore, phenylpropionate has a shorter period of action.
The increase in muscle mass occurs in smaller volumes and at a slower pace than when using Decanoate, but the process is more qualitative and lasts a longer period of time.
MEDICAL ACTION
Treatment with Fenandrol requires caution in (or a history of) heart disease and circulatory disorders, liver and kidney dysfunction, as well as epilepsy, migraine and glaucoma. During treatment with Fenandrol, it is recommended to check the intraocular pressure (for example eye pressure) regularly that there is a chance of retention of sodium and water. Hepatic impairment may occur, hepatic function testing should therefore be performed after 4 weeks of treatment. Before initiating the treatment and later on, prostate rectal control is recommended during the treatment. In diabetes, it is advisable to adjust the doses of anti-hyperglycaemic agents.
In patients with malignant tumors the duration of treatment will be determined individually and according to liver parameters.
SIDE EFFECTS
Like all medicines, this preparation can cause side effects, although not everybody gets them. Nandrolone injection in therapeutic doses is unlikely can cause side effects. During the prolonged treatment, the following side effects (frequency data not available) were observed: acne, hirsutism, male alopecia, irreversible thickening of the voice, dysmenorrhea, clitoral hypertrophy, gynecomastia and inhibit spermatogenesis, frequent erections and penis enlargement can be observed, libido change, nausea, vomiting, decreased appetite.
DOSAGE
Dosage with this drug occurs more often than Nandrolone Decanoate. The dose varies between 50-200 mg per injection, which in total is about 150-600 mg / week. Injections should be made every 2–3 days.


Roger –
I order in this store 3 times already. Good discounts today. All goods arrive on time. The quality is super. The store works fine, thanks